How To Draw Ionic Bonds
How To Draw Ionic Bonds - Web when drawing lewis dot structures for ionic compounds you need to follow a different set of rules than with lewis structures for covalent/molecular compounds. Magnesium now has an empty third shell so there is no need to draw a third. Draw a square bracket around each ion. Web the attraction of oppositely charged ions caused by electron transfer is called an ionic bond. The strength of ionic bonding depends on the magnitude of the charges and the sizes of the ions. Ionic bonds occur between metal ions and nonmetal ions, or polyatomic ions. In chapter 1, we used atomic theory to describe the structure of the fluorine atom. These grades are the stepping stone to your future. Web this chemistry video explains how to draw the lewis structures of ionic compounds. Web in ionic bonding, atoms transfer electrons to each other. Web the two types of bonding are covalent, for the sharing of electrons between atoms, and ionic, for the net transfer of electrons between atoms. Web draw a lewis electron dot diagram for an atom or a monatomic ion. The two electrons in the bond are simultaneously attracted to both nuclei. Web representing a covalent bond using lewis structures. In. In chapter 1, we used atomic theory to describe the structure of the fluorine atom. It has a giant lattice structure with strong electrostatic forces of attraction. Web in ionic bonding, atoms transfer electrons to each other. Web when drawing lewis dot structures for ionic compounds you need to follow a different set of rules than with lewis structures for. Covalent or ionic bonding will determine the type of compound that will be formed. In chapter 1, we used atomic theory to describe the structure of the fluorine atom. Magnesium loses two electrons and oxygen gains two electrons. In a covalent bond, the electrons are shared between atoms. If it gave away that electron it would become more stable. Web it's just for ionic compounds electrons aren't shared so you won't have things like single bonds between atoms. Ionic bonds require at least one electron donor and one electron acceptor. The strength of ionic bonding depends on the magnitude of the charges and the sizes of the ions. In electron transfer, the number of electrons lost must equal the. Web i want to help you achieve the grades you (and i) know you are capable of; Web draw a lewis electron dot diagram for an atom or a monatomic ion. 1.7.1 formation of covalent bonds; Web for exam purposes you need only show the outer electrons in dot & cross diagrams.you should be able to draw dot & cross. Web in ionic bonding, atoms transfer electrons to each other. Web when drawing lewis dot structures for ionic compounds you need to follow a different set of rules than with lewis structures for covalent/molecular compounds. Web it's just for ionic compounds electrons aren't shared so you won't have things like single bonds between atoms. Web the two types of bonding. The strength of ionic bonding depends on the magnitude of the charges and the sizes of the ions. If it could gain an electron from somewhere it too would become more stable. Sodium (2,8,1) has 1 electron more than a stable noble gas structure (2,8). Draw a square bracket around each ion. Covalent bonds occur mostly between nonmetal atoms. Web the attraction of oppositely charged ions caused by electron transfer is called an ionic bond. If it could gain an electron from somewhere it too would become more stable. The strength of ionic bonding depends on the magnitude of the charges and the sizes of the ions. Web this crash course chemistry video tutorial explains the main concepts between. In a covalent bond, the electrons are shared between atoms. Examples include nacl, mgf2, k2o, and al2o3. To facilitate our understanding of how valence electrons interact, a simple way of representing those valence electrons would be useful. This is because valence electrons. Even if you don't want to stud. Ionic bonds occur between metal ions and nonmetal ions, or polyatomic ions. Shows how to draw lewis dot structures for ionic compounds. Web representing a covalent bond using lewis structures. 2 show how electrons are transferred in ionic bonding. The strength of ionic bonding depends on the magnitude of the charges and the sizes of the ions. Web when drawing lewis dot structures for ionic compounds you need to follow a different set of rules than with lewis structures for covalent/molecular compounds. Web the attraction of oppositely charged ions caused by electron transfer is called an ionic bond. Web to begin our exploration of bonding, we need to define the two main types of bonds: Magnesium loses two electrons and oxygen gains two electrons. The two electrons in the bond are simultaneously attracted to both nuclei. Ionic bonds require at least one electron donor and one electron acceptor. It is assumed that only electrons in the valence shell are involved in the formation of covalent bonds. Ionic bonds occur between metal ions and nonmetal ions, or polyatomic ions. These structural diagrams depict only the outer, or valence, shell electrons and are known as dot and cross diagrams. Web this crash course chemistry video tutorial explains the main concepts between ionic bonds found in ionic compounds and polar & nonpolar covalent bonding foun. Web 6.7k views 7 years ago edexcel chemistry 2022: Shows how to draw lewis dot structures for ionic compounds. In contrast, atoms with the same electronegativity share electrons in covalent bonds, because neither atom preferentially attracts or repels the shared electrons. Web draw a lewis electron dot diagram for an atom or a monatomic ion. To facilitate our understanding of how valence electrons interact, a simple way of representing those valence electrons would be useful. The strength of ionic bonding depends on the magnitude of the charges and the sizes of the ions.
How Do Ions Increase Conductivity? Atlas Scientific
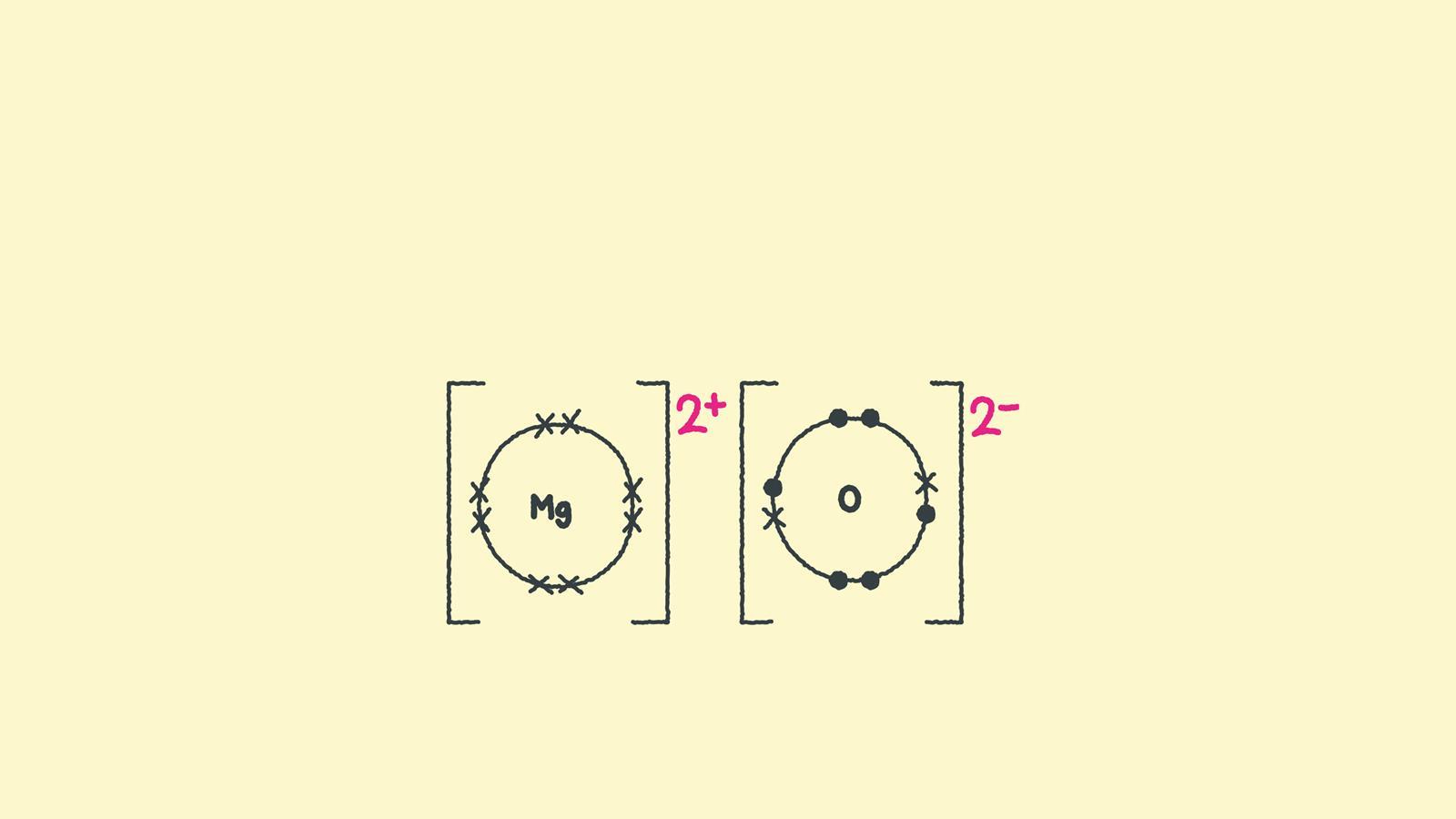
How to draw ionic bonding dot and cross diagrams Feature RSC Education
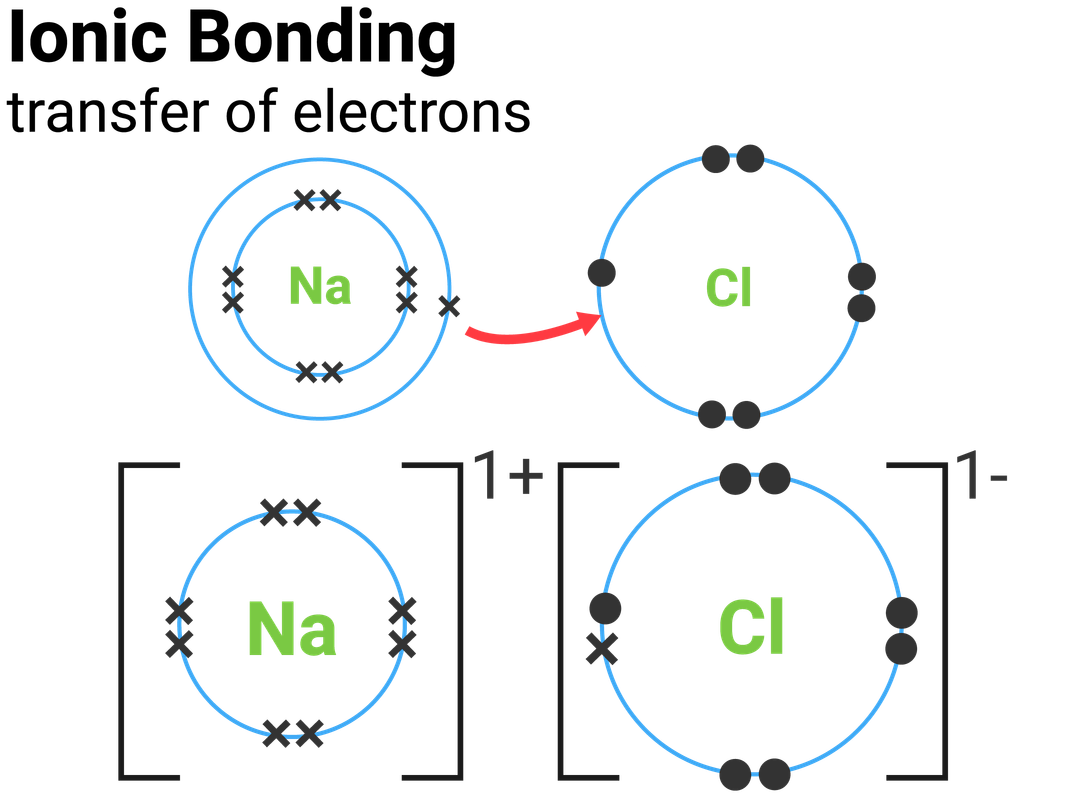
6 ionic bonding 01 — Postimages
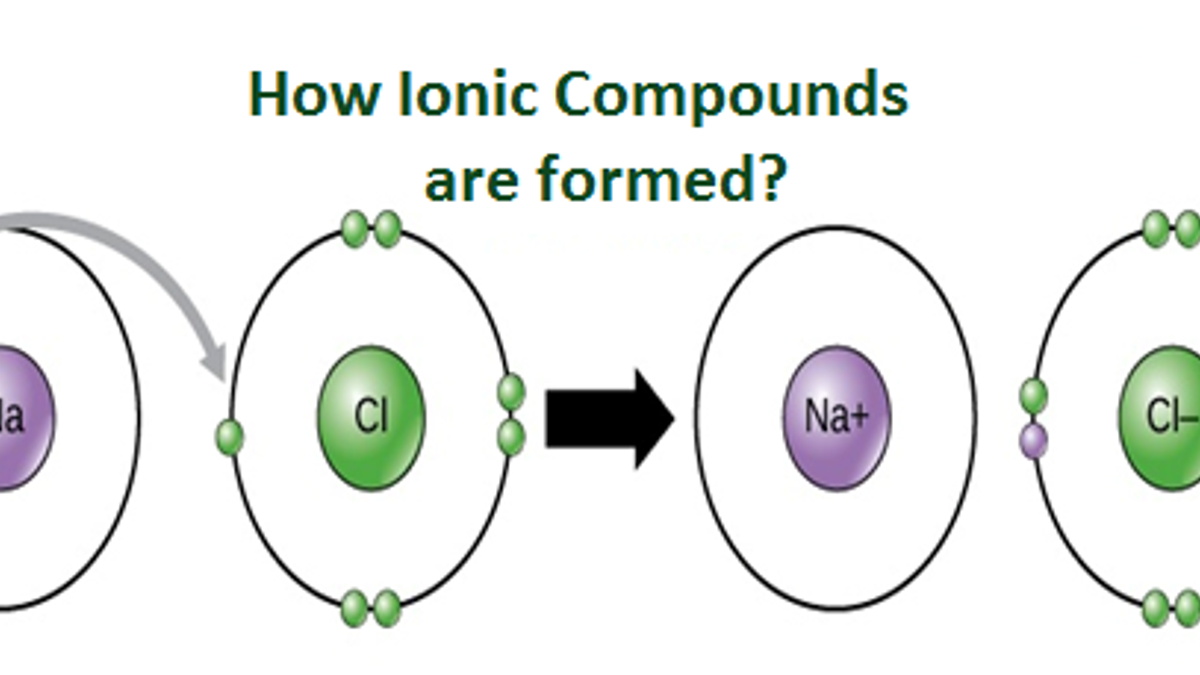
How Ions Are Formed? Infrared for Health

Lewis Structure Of Ionic Compounds
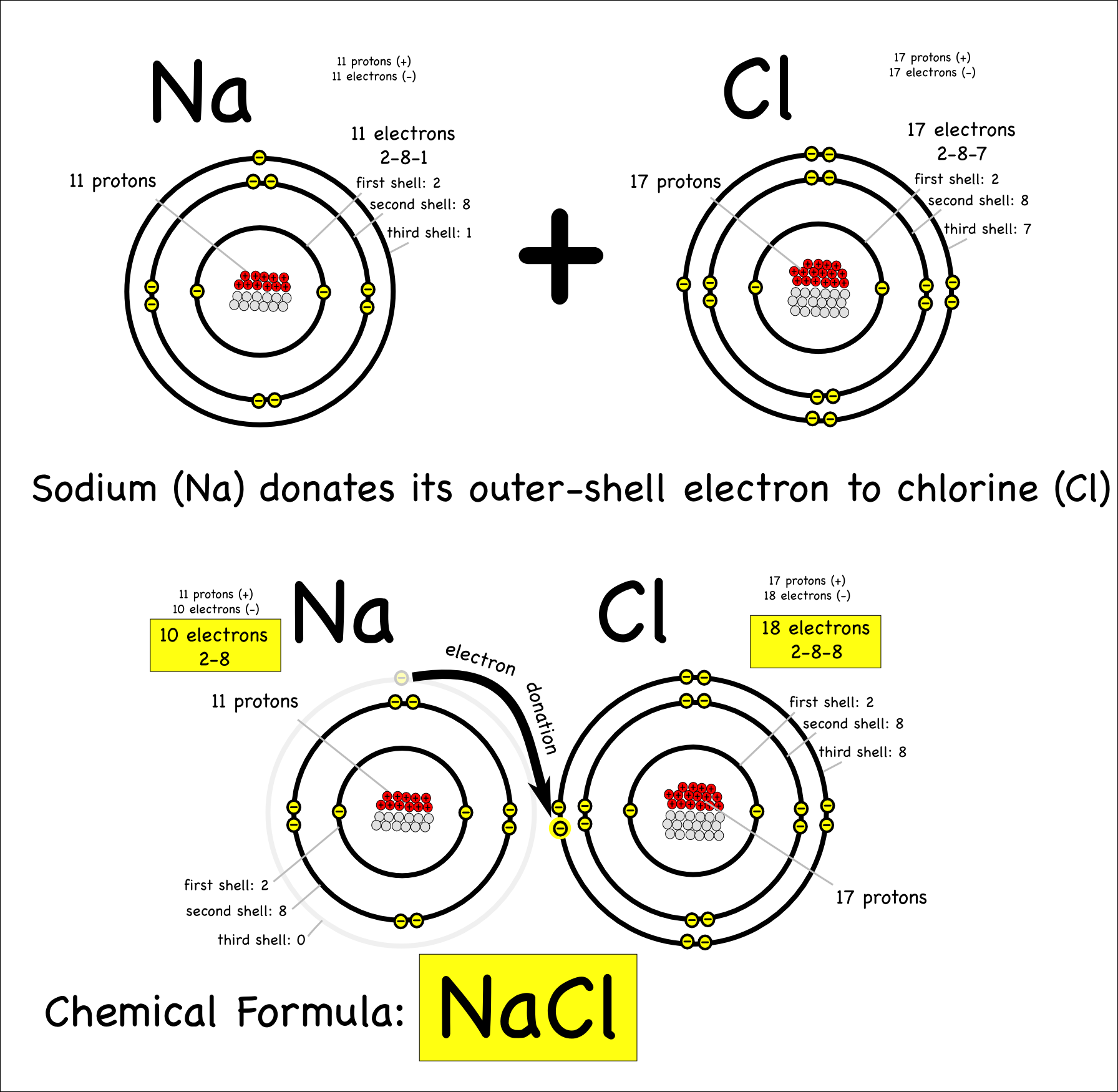
subatomic particles Montessori Muddle
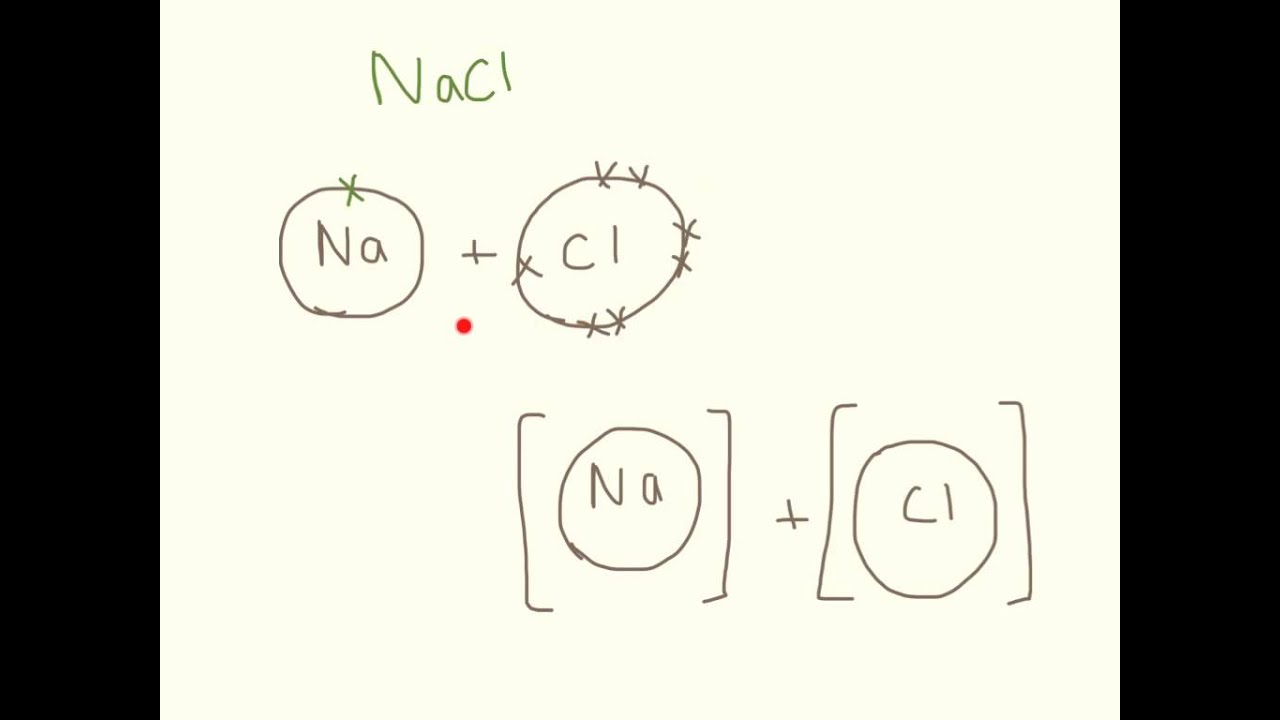
drawing ionic bonds worksheet lineartdrawingsheartflowers
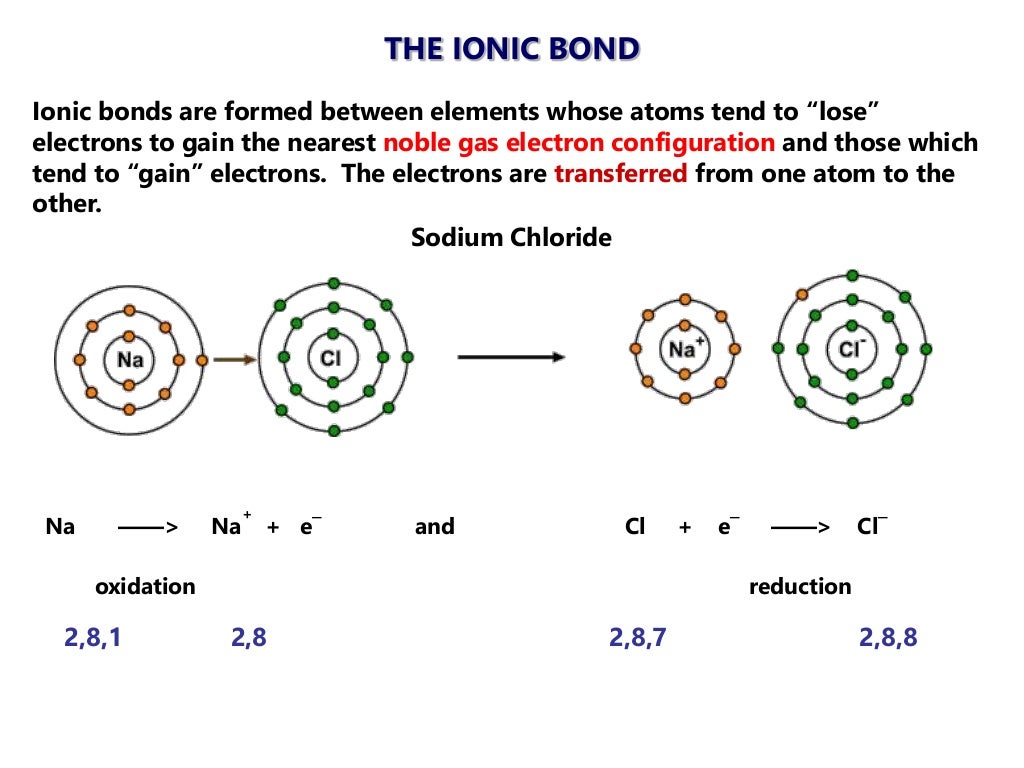
2016 topic 4.1 bonding ionic
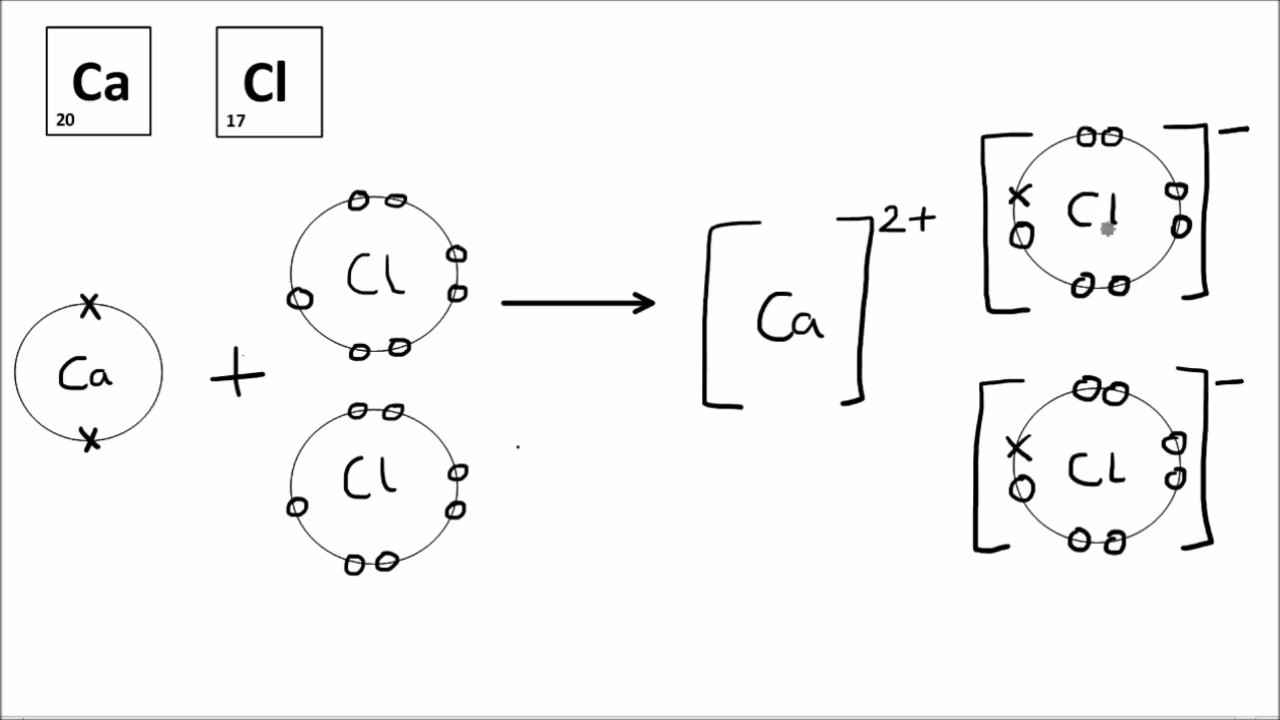
Ionic Bonding Diagram
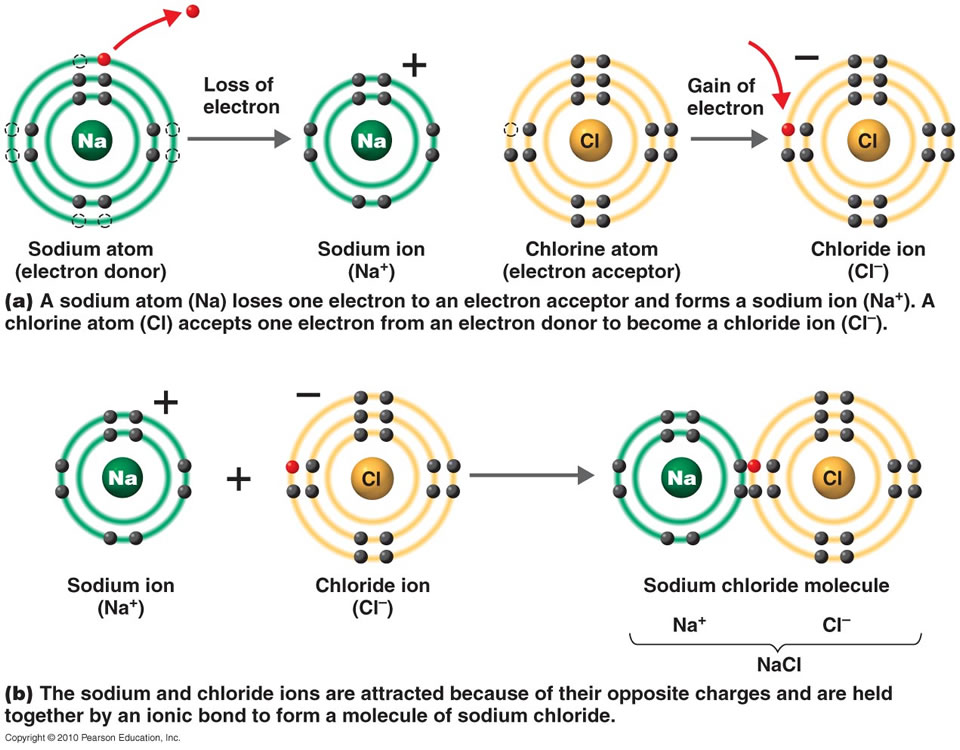
savvychemist Ionic Bonding (2) Dot and cross diagrams/Lewis structures
In A Covalent Bond, The Electrons Are Shared Between Atoms.
In Electron Transfer, The Number Of Electrons Lost Must Equal The Number Of Electrons Gained.
It Has A Giant Lattice Structure With Strong Electrostatic Forces Of Attraction.
Web It's Just For Ionic Compounds Electrons Aren't Shared So You Won't Have Things Like Single Bonds Between Atoms.
Related Post: