Draw The Main Lewis Structure Of Nof
Draw The Main Lewis Structure Of Nof - Web the lewis structure of nof is drawn by first tallying up the total number of valence electrons from all the atoms, which in this case is 5 from nitrogen, 6 from oxygen, and 7 from fluoride (total 18 electrons). Web this video illustrates the thinking behind determining the lewis structure of a simple molecule and using that information to determine the electron pair and. Calculate the total number of valence electrons. Check the formal charges to be sure that each atom has a formal charge of zero. It was introduced in 1916 by gilbert n. Draw nonbonding electrons using the dot notation and bonding electrons as a bond. Web the first step is to draw the lewis structure of the molecule. Draw the main lewis structure of nof. Nof is actually onf, since. This problem has been solved! Web the lewis structure of nof is drawn by first tallying up the total number of valence electrons from all the atoms, which in this case is 5 from nitrogen, 6 from oxygen, and 7 from fluoride (total 18 electrons). First determine the total number of valence electrons in the molecule. By using the following steps, you can easily draw. For the nof structure use the periodic table to find the total number. Web chemistry questions and answers. Determine the number of bonding electrons and the number of nonbonding electrons in. #1 first draw a rough sketch. #5 repeat step 4 if needed, until all charges are minimized, to get a stable lewis structure. Include all lone pairs of electrons. View available hint (s) ννου ch on spf brax more. The lewis structure for nof is: Web 453 views 1 year ago lewis structure. Web chemistry questions and answers. #4 convert lone pairs of the atoms, and minimize formal charges. When drawing the structure of an ion, be sure to add/subtract electrons to account for. For \(\ce{pcl3}\), the electron dot diagram is as follows: Draw the main lewis structure of nof. And to help you understand the lewis structure of this molecule, we are going to share our. Draw the main lewis structure of nof. Draw the main lewis structure of nof. Web chemistry questions and answers. Web 453 views 1 year ago lewis structure. Draw nonbonding electrons using the dot notation and bonding electrons as a bond. Nof is actually onf, since. Draw the lewis structure for nof. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. In the nof lewis structure nitrogen (n) is the least electronegative atom and goes in the center of the lewis structure. For pcl 3, the electron dot diagram is as follows: For the nof structure use the periodic table to find the total number. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Nitrogen is the central atom. Here, the given molecule is nof. #3 calculate and mark formal charges on the atoms, if required. For the nof structure use the periodic table to find the total number. #5 repeat step 4 if needed, until all charges are minimized, to get a stable lewis structure. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. In order to draw the lewis structure of nof, first of all you have. Include all lone pairs of electrons. And to help you understand the lewis structure of this molecule, we are going to share our. Nitrogen is the central atom. #5 repeat step 4 if needed, until all charges are minimized, to get a stable lewis structure. Web the lewis structure is based on the octet rule, which states that atoms tend. It was introduced in 1916 by gilbert n. Draw nonbonding electrons using the dot notation and bonding electrons as a bond. Part b draw the main lewis structure of nof. Web steps of drawing nof lewis structure step 1: Web chemistry questions and answers. Draw the main lewis structure of nof. #5 repeat step 4 if needed, until all charges are minimized, to get a stable lewis structure. Web the nof lewis structure is very similar to nocl and nobr. The central atom is typically the least electronegative, which is nitrogen. And to help you understand the lewis structure of this molecule, we are going to share our. Web use these steps to correctly draw the nof lewis structure: Web how to draw lewis diagrams. #3 calculate and mark formal charges on the atoms, if required. #1 first draw a rough sketch. Draw nonbonding electrons using the dot notation and bonding electrons as a bond. B) draw the lewis structure for nof (nitrogen is the central atom) this problem has been solved! Web the first step is to draw the lewis structure of the molecule. In order to find the total valence electrons in a nof molecule, first of all you should know the valence electrons present in nitrogen atom, oxygen atom as well as fluorine atom. #2 mark lone pairs on the atoms. First determine the total number of valence electrons in the molecule. Draw the main lewis structure of nof.
Draw The Main Lewis Structure Of Nof.
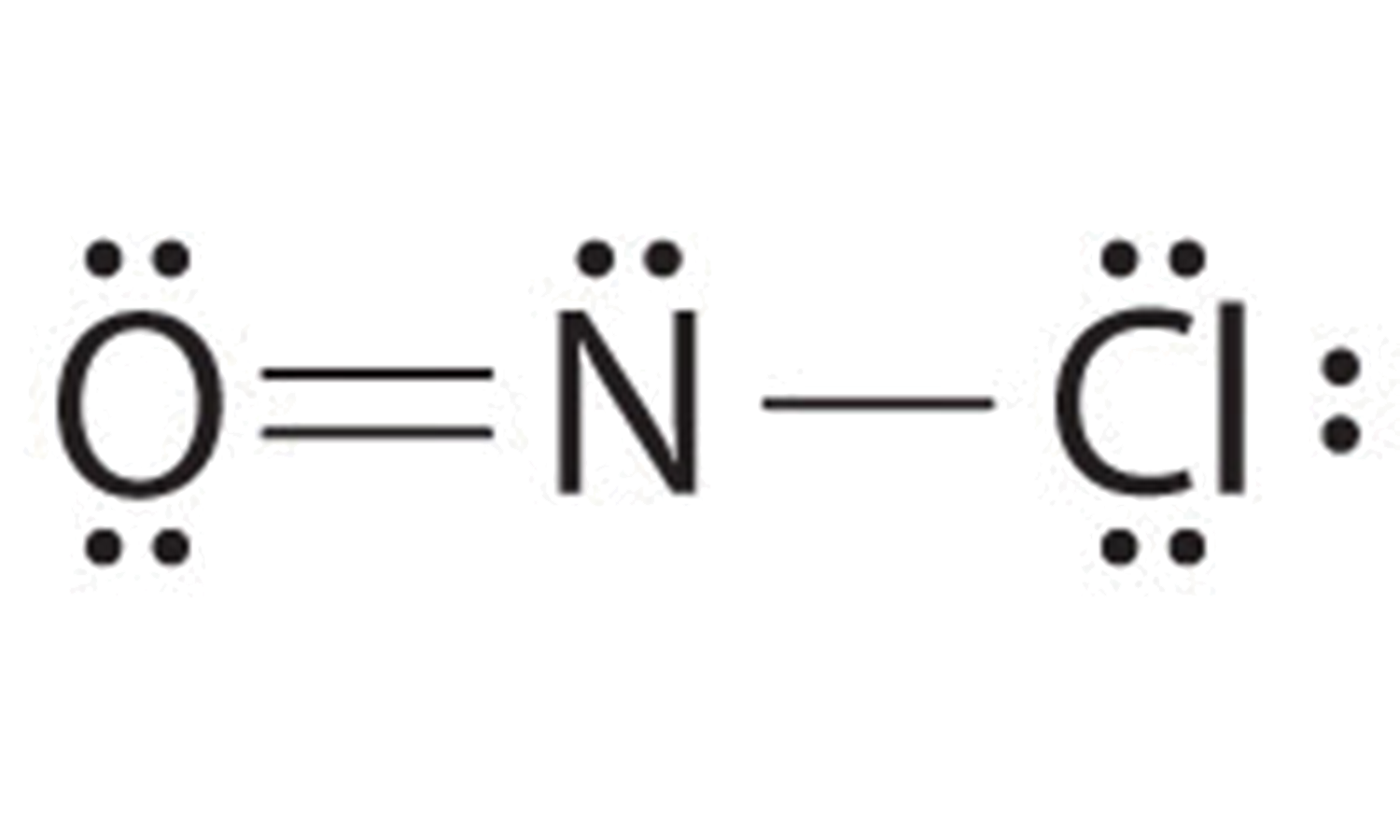
Main Lewis Structure Of Nof

Structure and Geometry The NOF example YouTube

NOF Lewis structure, molecular geometry, bond angles, resonance

Nof Lewis Structure With Charges

NOF Lewis Structure, Geometry, Hybridization, and Polarity

Draw the main lewis structure of nof. draw nonbonding electrons using
Solved Part B Draw the main Lewis structure of NOF. Draw

NOF Lewis Structure How to Draw the Lewis Structure for NOF YouTube

How to draw lewis structures for NOF in 60s! Dr K shorts YouTube
Web Draw The Lewis Structure For Nof Nitrogen Is The Central Atom Draw The Molecule By Placing Atoms On The Grid And Connecting Them With Bonds.
By Using The Following Steps, You Can Easily Draw The Lewis Structure Of Nof.
Web The Lewis Structure Of Nof Is Drawn By First Tallying Up The Total Number Of Valence Electrons From All The Atoms, Which In This Case Is 5 From Nitrogen, 6 From Oxygen, And 7 From Fluoride (Total 18 Electrons).
#5 Calculate Formal Charge And Check Stability.
Related Post: